A case of stents, missed lesions and congestion of the spinal veins
Recently, a patient wanted to know why I had changed my position on stenting the internal jugular veins. I was surprised, because I remain conservative in their use but acknowledge that stents can be helpful and safe provided they are deployed safely and the post-stenting period is managed properly with surveillance and anticoagulation
.
Perhaps the reasons are related to my recent more frequent encounters with patients who have already had multiple procedures.
I find it uncommon for patients to require stents during the first treatment. Most of the time, the stenoses, commonly caused by immobile valves, are readily opened by use of high pressure fairly large balloons.
The patient about whom I write is a 37 year old female who was diagnosed with relapsing remitting multiple sclerosis in 2004. She remains in the relapsing for , having had three exacerbations in the past two years.
Her major disability was weakness in her right lower extremity. This was associated with quite significant spasticity, rendering the knee quite inflexible. The weakness was most pronounced more proximally. Hip flexors and knee extenders was weak bilaterally, but with more weakness on the right side. ,
Sensory deficits included numbness in both lower extremities and the right hand.
She also suffered from fatigue and intolerance to heat. Bladder urgency was also present.
She had two prior endovascular procedures for CCSVI before I met her.
Her first treatment was in summer 2009. Narrowings were seen in the J-3 segment of each jugular vein. These were treated by balloon angioplasty and self-expanding 12 mm stents. Significant pain was present for about three months from these high stents. However there were no complications such as vagal or spinal accessory neuropraxia. No thrombosis was ever described. She had dramatic improvements in her bladder urgency and in her spasms. However after conception of her first child, these benefits regress.
She had another venogram with IVUS in 2011. At that time both internal jugular veins underwent double balloon angioplasty . Some treatment of the azygous vein was also undertaken. The entire procedure was said to have lasted about 30 minutes. This intervention led to return of improvefments in urinary symptoms and muscle spasm. Unfortunately, her gains were lost during her second pregnancy.
During her first procedure, stents had been placed in both internal jugular veins in the J-3 segments. These were 12 mm in diameter. The patients reports excrutiating pain from the stents that lasted several months. She did not report any shoulder weakness (CN11), hoarseness (CN 10) or other symptoms.
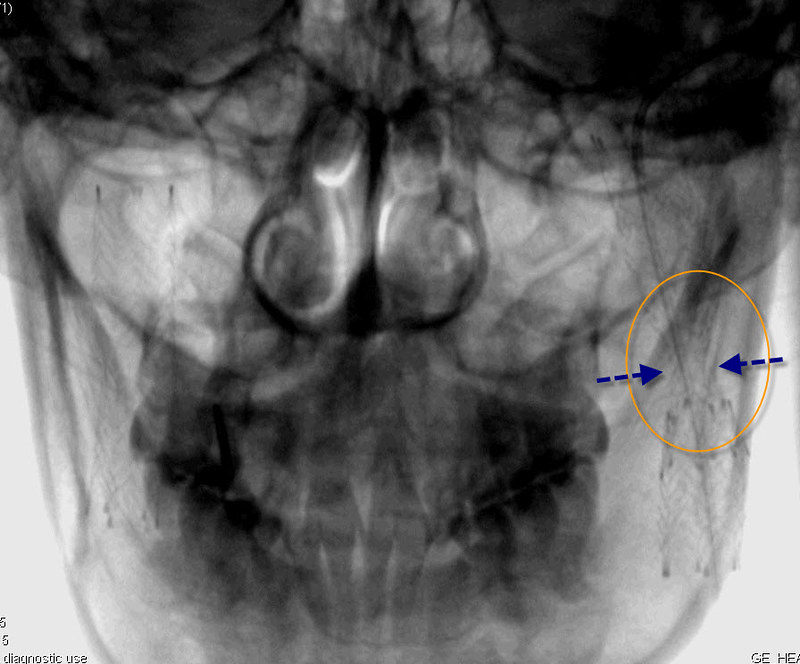
The Lower left stent (orange circle) had concentric narrowing on the frontal view (blue arrows), but was not narrowed on the lateral view.
I have a conservative viewpoint on the utilization of stents in the jugular veins. I believe they have a purpose and can salvage of poor clinical outcome in the right circumstance. However, I rarely use them and only in specific indications. My indications for stenting the internal jugular vein are in two categories: acute bale-out procedures, and problems related to restenosis.
Bale out procedures are sometimes necessary to correct complications of venoplasty. Intimal flaps may occur during angioplasty, either caused by too large a balloon, by too tight attachments of valves and septa to the vein wall and by unfavorable angle of angioplasty. Sometimes the created flap protrudes into the lumen of the vein; it can become occlusive and result in thrombosis. Stents are useful in keeping the vein open in this situation. In other occasions patients present with a chronically thrombosed vein from a prior treatment. Re-establishing flow requires that we to navigate the guidewire and catheter across the thrombosis into the non-clotted vein above. Angioplasty alone is unlikely to maintain blood flow as thrombosiscommonly recurs. Stenting may provide a way of keeping some of these thrombosed veins open.
Other indications for stenting are related to repeated failures of angioplasty in patients who have demonstrated some (usually short-term) benefit. These repeat obstructions are often the result of recurrent stenosis due to elastic recoil or anatomical problems that cause the veins to repeatedly become obstructed. Other indications for stenting, such as tumor compressions, radiation or surgical scarring, are uncommonly seen in patients with MS. None of the other indications are used during my initial treatment. They are reserved for conditions where it is clear that simple angioplasty is not going to result in a durable long lasting clinical improvement.
I discuss with my patients both of these scenarios but my patients know and are prepared for the use of stents in bale-out procedures. The other indications, such as resistant or recurrent stenoses and other problems can be further discussed and decided, but not while the patient is in the midst of a procedure, for they rarely require urgent decisions.
Why such cautious use of stents? Because they are not without risks. Undersizing stents or unexpected distension of the vein may lead to migration. Thrombosis can occur. Intimal hyperplasia may result in in-stent stenosis.
Cutaneous and systemic hypersensitivity reactions to implanted metals are of concern although the incidence and prevalence of such hypersensitivity reactions are unknown. In the past, such allergic reactions were not considered to be common. But more recent case series have suggested that such reactions are not uncommon and may be greater than 5% of patients with implantations. Thus placing stents in a non-urgent situation without evaluating hypersensitivity evaluation my be unwise. Those patients with a reported history of metal dermatitis should be evaluated by patch testing. Those without a history of dermatitis should not be tested unless considerable concern exists.
Overall, it seems that metal allergy is not a risk factor for In-stent restenosis (ISR) (following stenting with stainless steel stents). However, nickel allergies causing ISR have not been conclusively disproved.
I present to you a case of long standing (greater than three years) stenting. While this review of the stents is interesting, other concepts are also reviewed in this case.
The patient about whom I write is a 37 year old female who was diagnosed with relapsing remitting multiple sclerosis in 2004. She remains in the relapsing remitting form, having had three exacerbations in the past two years.
Her major disability is weakness in her right lower extremity, associated with quite significant spasticity, rendering the knee quite inflexible. The weakness is most pronounced more proximally. Hip flexors and knee extenders are weak bilaterally, but with more weakness on the right side. Sensory deficits include numbness in both lower extremities and the right hand. She also has fatigue and intolerance to heat. Bladder urgency is also present, although it is better than before the first procedure.
She had two prior endovascular procedures for CCSVI before I met her.
Her first treatment was in summer 2009. Narrowings were seen in the J-3 segment of each jugular vein. These were treated by balloon angioplasty and self-expanding 12 mm stents. Significant pain was present for about three months from these high stents. However there were no complications such as vagal or spinal accessory neuropraxia. No thrombosis was ever described. She had dramatic improvements in her bladder urgency and in her spasms. However after conception of her first child, these benefits regress.
She had another venogram with IVUS in 2011. At that time both internal jugular veins underwent "kissing" balloon angioplasty. Some treatment of the azygous vein was also undertaken. The entire procedure was said to have lasted about 30 minutes. This intervention led to lesser improvefments in urinary symptoms and muscle spasm. Unfortunately, her gains were lost during her second pregnancy.
Her ultrasound examination revealed that there were three criteria fulfilled for sonography diagnosis of CCSVI, namely reflux in the jugular vein in supine and erect positions, reflux in the deep cerebral veins in any position and B-mode abnormalities including a possible septum on the right jugular, immobile valves in both jugular veins.
I started with right dural sinus and internal jugular venography.
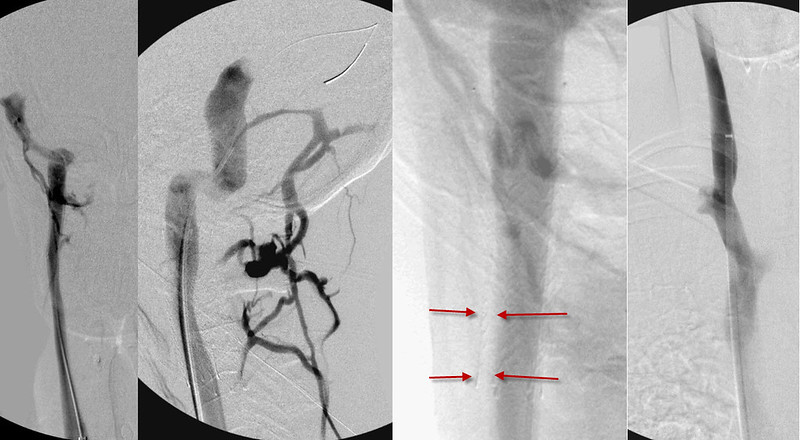
There was reflux into the mastoid emissary vein and posterior cervical veins and in-stent stenosis (red arrows) narrowing slightly the luminal column inside the stent. Extra contrast puddles were noted on the lateral side of the jugular vein at the inferior bulb.
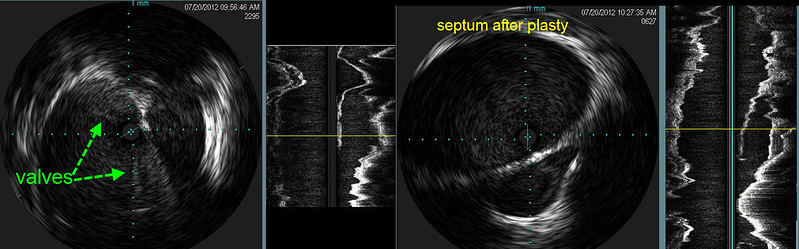
IVUS before (left 2 images) showed that there were immobile incompletely opening thickened valves (green arrows) and a fluid containing area to the right of the jugular vein. The next image is a longitudinal view and one can clearly see the blind ending sac outlined by thickened tissue representing a septum. IVUS after angioplasty (right 2 images) showed that the valve is opened but there is no change in the septum.
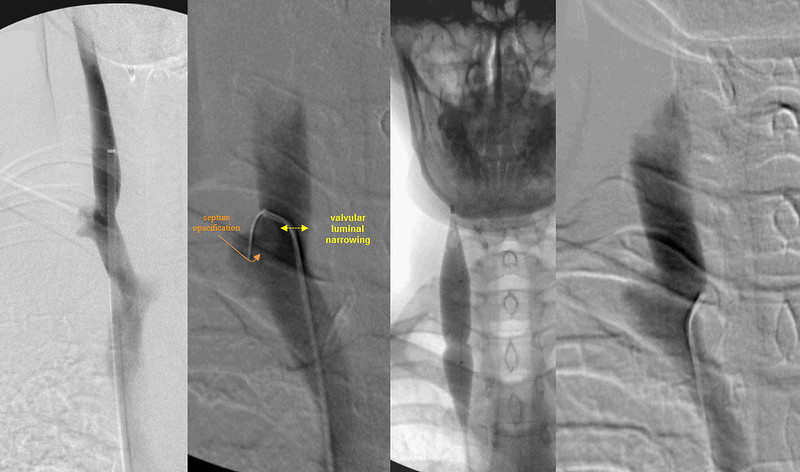
The outpocketing of contrast media to the left of the internal jugular vein represents contrast extending into the septum. To prove it I placed a catheter into the orifice of this septum (orange arrow). After balloon angioplasty the contrast column is smoother and the flow was faster but the bulge has changed little.
Next left internal jugular and dural sinus venography were performed.
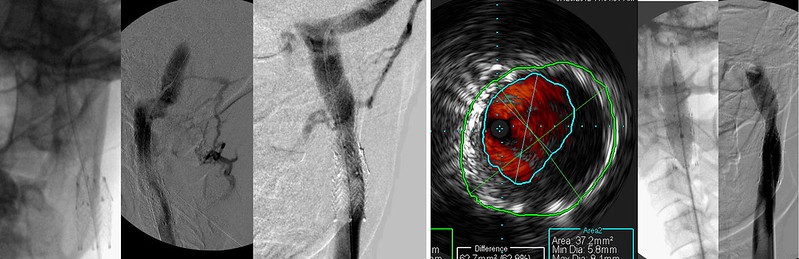
Examination of the stents by contrast study of the dural sinus (image 2,3) showed that there was narrowing and irregularity of the contrast column as it traversed the stent.
IVUS (image 4) shows that there is in-stent thickening. The outer green line outlines the stent. The blue line outlines the stent flow (represented in red). Balloon angioplasty (image5) was performed. There was improved flow although some stenosis remains. Note that the contrast column is now smooth, fills the stent with no irregularities, only a focal stenosis. This corresponds to the narrowed stent, presumably caused by bony compression.
The remainder of the left internal jugular vein was then imaged.
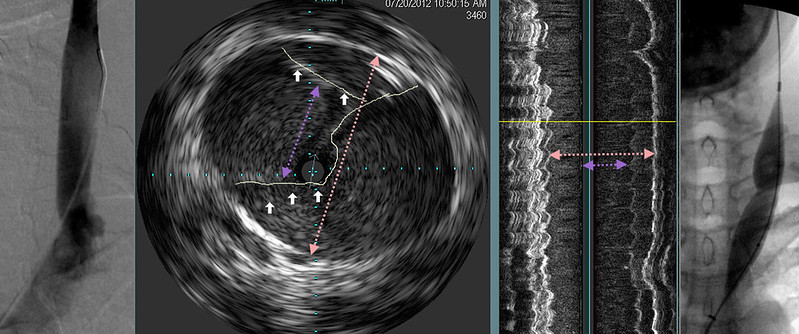
At the inferior jugular bulb stenosis was seen on venography. The edges of the valves have been outlined by white lines. One can see that the diameter of the vein is much larger than the narrowed oifice outlined by valves. This stenosis is correlated with a longitudinal image. The valve area is clearly narrowed.
Angioplasty confirmed the stenosis.
The azygous vein was also abnormal.
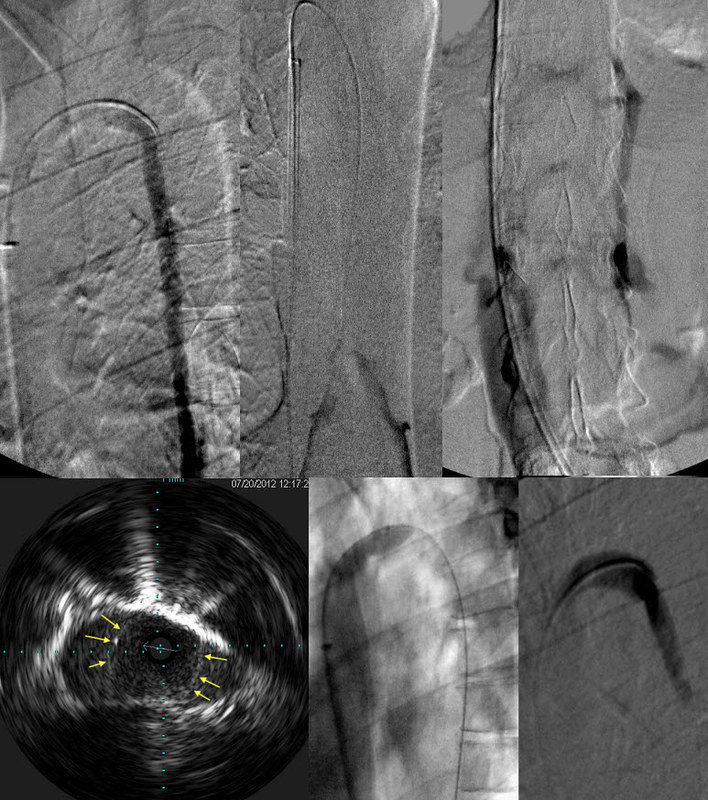
TOP ROW
With the catheter injection at the junction of the azygous arch and the ascending azygous vein, one sees NO flow of contrast reaching the superior vena cava; rather there is only retrograde flow down into the abdomen. .
The second image shows a contrast injection of the peripheral ascending azygous vein. There is reflux from confluens of azygous and hemiazygous veins with NO contrast flowing toward the superior venacava.
Finally, the third image shows reflux into the intervertebral plexus and the inferior vena cava.
OUT FLOW obstruction of the azygous vein is a problem here.
BOTTOM ROW
IVUS finds the lesion; an incomplete immobile valve in the arch of the azygous vein. (arrows outline the immobile valve). This was treated by angioplasty (middle). The resulting flow was strong and no reflux was seen. (right)

The azygous vein is only one part of this patient's thoracoabdominal plethora. In addition to the azygous disease seen on the first three panels of this image, there is also a hypoplastic ascending lumbar vein draining the lumbar spine in the fourth panel. This vein generally drains into the renal vein. The vein shows numerous areas where there are attenuated lumbar branches. The fifth panel shows evidence of a Nutcracker phenomenon with reflux into the ovarian vein and the ascending lumbar vein. Thus cerebrospinal drainage is made worse by the outflow obstruction of the renal vein's 550 ml/min flow seeking alternate pathways to the heart.
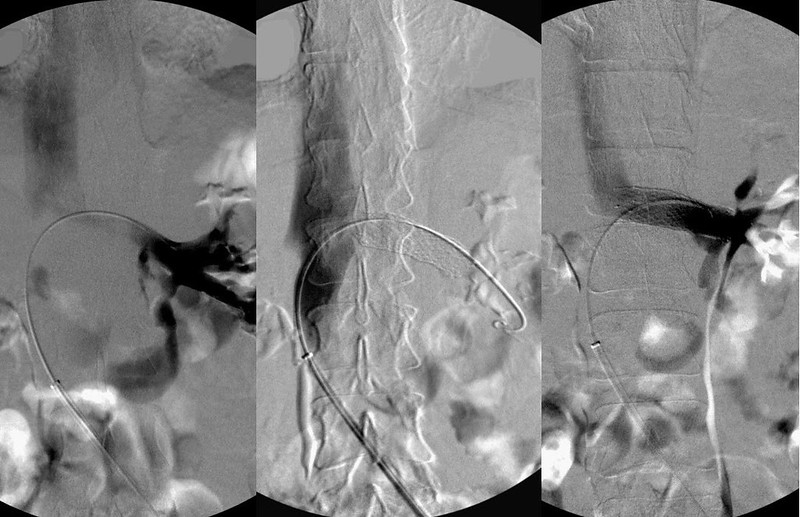
Nutcracker phenomenon results from compression of the left renal vein between the abdominal aorta and the superior mesenteric vein. The Left renal vein flows at about 550 ml/minute, which is 2-3 times the flow of both azygous and ascending lumbar veins combined. With outflow obstructions of the azygous and the ascending lumbar vein outflow, one can readily recognize the consequences of the renal vein obstruction as it converts the renal vein into an inflow vein of the cerebrospinal venous bed.
Treatment of this compressive obstruction is stenting. (image 3). Angioplasty alone is ineffective in reversing the renal vein compression.
Unlike the jugular vein which is stented with great reluctance, stenting of the renal vein is the only method short of abdominal exploration and reattachment of the renal vein.
Now we will wait to see if the patient's symptoms will improvef. I am always skeptical of possible improvements in strong spasticity. I think of this as a quintessential MS symptom. While I have seen improvements in spasticity, resolution of it does not reach the petential for improvements like those of loss of balance, cog fog and fatigue
Learning points
1.Stents can remain patent for many years without migration but with some compression. Stenosis can be both in-stent restenosis and external compression, as we have seen today.
2. Periodic management of these in-stent restenoses is relatively simple.
3. It took three venograms and two IVUS before an azygous arch stenosis was discovered.
4. A nutcracker syndrome was not considered by two prior angiographers.
5. We have seen and recognized the mastoid emissary veins for the first time
6. Septums are very difficult to treat. Balloon angioplasty just compresses it back up against the wall of the vein.



 The outpocketing of contrast media to the left of the internal jugular vein represents contrast extending into the septum. To prove it I placed a catheter into the orifice of this septum (orange arrow). After balloon angioplasty the contrast column is smoother and the flow was faster but the bulge has changed little.
The outpocketing of contrast media to the left of the internal jugular vein represents contrast extending into the septum. To prove it I placed a catheter into the orifice of this septum (orange arrow). After balloon angioplasty the contrast column is smoother and the flow was faster but the bulge has changed little. 



