NAD+ for mitochondria
NAD+ for mitochondria
I ran across a supplement the other day that contains nicotinamide riboside which goes by the trade name of Niagen. It's supposed to help bump up mitochondrial activity by increasing NAD+ levels. Could this be the missing link for reactivating mitochondria in MS and possibly slowing or preventing neurodegeneration?
http://www.lifeextension.com/Magazine/2 ... AD/Page-01
Note that Dr. Ben Lynch also sells a NADH supplement.
http://www.seekinghealth.com/nadh-coq10 ... ealth.html
More info from Chromadex the company that makes Niagen.
https://chromadex.com/Ingredients/NIAGEN.html
Question: Has anybody tried this and what were the results?
http://www.lifeextension.com/Magazine/2 ... AD/Page-01
Note that Dr. Ben Lynch also sells a NADH supplement.
http://www.seekinghealth.com/nadh-coq10 ... ealth.html
More info from Chromadex the company that makes Niagen.
https://chromadex.com/Ingredients/NIAGEN.html
Question: Has anybody tried this and what were the results?
- CureOrBust
- Family Elder
- Posts: 3374
- Joined: Wed Jul 27, 2005 2:00 pm
- Location: Sydney, Australia
Re: NAD+ for mitochondria
I did a quick search, but could not find any direct information regarding if this product readily crosses the BBB?
I also came across a brief basically unsupported statement that it functions to replace the action of B3, in a non-flushing form. I do know of Picamilon, which is a non flushing form of B3 that is specifically known to cross the BBB. However, I have no idea on its effects on NAD+ A lot of the links I followed, appeared to be mostly effects on NAD+ outside of the CNS.
B3 & NAD+ (Haven't read the page myself)
http://www.longecity.org/forum/topic/72 ... ce-thread/
Picamilon in the Wiki
https://en.wikipedia.org/wiki/Picamilon
I also came across a brief basically unsupported statement that it functions to replace the action of B3, in a non-flushing form. I do know of Picamilon, which is a non flushing form of B3 that is specifically known to cross the BBB. However, I have no idea on its effects on NAD+ A lot of the links I followed, appeared to be mostly effects on NAD+ outside of the CNS.
B3 & NAD+ (Haven't read the page myself)
http://www.longecity.org/forum/topic/72 ... ce-thread/
Picamilon in the Wiki
https://en.wikipedia.org/wiki/Picamilon
Re: NAD+ for mitochondria
I found another brand on Amazon which appears to be the same formulation (the tablets even look the same) and on a $/mg basis it's about 1/3 less.NHE wrote:Note that Dr. Ben Lynch also sells a NADH supplement.
http://www.seekinghealth.com/nadh-coq10 ... ealth.html
http://www.amazon.com/Fatigue-Naturally ... B011MZORKU
Here's the main site for this company and the NADH+CoQ10 appears to be their only product.
http://www.maac10.com/
Re: NAD+ for mitochondria
Here are a couple of interesting papers...
Nicotinamide riboside restores cognition through an upregulation of proliferator-activated receptor-γ coactivator 1α regulated β-secretase 1 degradation and mitochondrial gene expression in Alzheimer's mouse models.
Neurobiol Aging. 2013 Jun;34(6):1581-8.
Effective treatment of mitochondrial myopathy by nicotinamide riboside, a vitamin B3.
EMBO Mol Med. 2014 Apr 6;6(6):721-31.
Partial reversal of skeletal muscle aging by restoration of normal NAD⁺ levels.
Rejuvenation Res. 2014 Feb;17(1):62-9.
Nicotinamide riboside restores cognition through an upregulation of proliferator-activated receptor-γ coactivator 1α regulated β-secretase 1 degradation and mitochondrial gene expression in Alzheimer's mouse models.
Neurobiol Aging. 2013 Jun;34(6):1581-8.
- Nicotinamide adenine dinucleotide (NAD)(+), a coenzyme involved in redox activities in the mitochondrial electron transport chain, has been identified as a key regulator of the lifespan-extending effects, and the activation of NAD(+) expression has been linked with a decrease in beta-amyloid (Aβ) toxicity in Alzheimer's disease (AD). Nicotinamide riboside (NR) is a NAD(+) precursor, it promotes peroxisome proliferator-activated receptor-γ coactivator 1 (PGC)-1α expression in the brain. Evidence has shown that PGC-1α is a crucial regulator of Aβ generation because it affects β-secretase (BACE1) degradation. In this study we tested the hypothesis that NR treatment in an AD mouse model could attenuate Aβ toxicity through the activation of PGC-1α-mediated BACE1 degradation. Using the Tg2576 AD mouse model, using in vivo behavioral analyses, biochemistry assays, small hairpin RNA (shRNA) gene silencing and electrophysiological recording, we found (1) dietary treatment of Tg2576 mice with 250 mg/kg/day of NR for 3 months significantly attenuates cognitive deterioration in Tg2576 mice and coincides with an increase in the steady-state levels of NAD(+) in the cerebral cortex; (2) application of NR to hippocampal slices (10 μM) for 4 hours abolishes the deficits in long-term potentiation recorded in the CA1 region of Tg2576 mice; (3) NR treatment promotes PGC-1α expression in the brain coinciding with enhanced degradation of BACE1 and the reduction of Aβ production in Tg2576 mice. Further in vitro studies confirmed that BACE1 protein content is decreased by NR treatment in primary neuronal cultures derived from Tg2576 embryos, in which BACE1 degradation was prevented by PGC-1α-shRNA gene silencing; and (4) NR treatment and PGC-1α overexpression enhance BACE1 ubiquitination and proteasomal degradation. Our studies suggest that dietary treatment with NR might benefit AD cognitive function and synaptic plasticity, in part by promoting PGC-1α-mediated BACE1 ubiquitination and degradation, thus preventing Aβ production in the brain.
Free full text.
Effective treatment of mitochondrial myopathy by nicotinamide riboside, a vitamin B3.
EMBO Mol Med. 2014 Apr 6;6(6):721-31.
- Nutrient availability is the major regulator of life and reproduction, and a complex cellular signaling network has evolved to adapt organisms to fasting. These sensor pathways monitor cellular energy metabolism, especially mitochondrial ATP production and NAD(+)/NADH ratio, as major signals for nutritional state. We hypothesized that these signals would be modified by mitochondrial respiratory chain disease, because of inefficient NADH utilization and ATP production. Oral administration of nicotinamide riboside (NR), a vitamin B3 and NAD(+) precursor, was previously shown to boost NAD(+) levels in mice and to induce mitochondrial biogenesis. Here, we treated mitochondrial myopathy mice with NR. This vitamin effectively delayed early- and late-stage disease progression, by robustly inducing mitochondrial biogenesis in skeletal muscle and brown adipose tissue, preventing mitochondrial ultrastructure abnormalities and mtDNA deletion formation. NR further stimulated mitochondrial unfolded protein response, suggesting its protective role in mitochondrial disease. These results indicate that NR and strategies boosting NAD(+) levels are a promising treatment strategy for mitochondrial myopathy.
Free full text.
Partial reversal of skeletal muscle aging by restoration of normal NAD⁺ levels.
Rejuvenation Res. 2014 Feb;17(1):62-9.
- That some aging-associated phenotypes may be reversible is an emerging theme in contemporary aging research. Gomes et al. report that age-associated oxidative phosphorylation (OXPHOS) defects in murine skeletal muscle are biphasic. In the first phase, OXPHOS is decreased because of reduced expression of mitochondrially encoded genes. Treatment of moderately old mice (first-phase OXPHOS defects) with nicotinamide adenine dinucleotide (NAD⁺) precursor nicotinamide mononucleotide (NMN) for 1 week restores oxidative phosphorylation activity and other markers of mitochondrial function in skeletal muscle. However, muscle strength is not restored. In very old animals (second-phase OXPHOS defects), expression of OXPHOS genes from both the nucleus and mitochondria is reduced and mitochondrial DNA integrity is diminished. Gomes et al. propose a model linking decreased NAD⁺ to loss of nuclear SIRT1 activity to stabilization of the hypoxia-associated transcription factor hypoxia-inducible factor 1-alpha (HIF-1a). HIF-1a promotes an hypoxic-like (Warburg effect) state in the cell. The HIF-1a protein interacts with c-Myc, decreasing c-Myc-regulated transcription of the key mitochondrial regulator mitochondrial transcription factor A (TFAM). Low levels of TFAM lead to first-phase OXPHOS dysfunction. The transition to irreversible phase 2 dysfunction remains to be characterized, but may be related to increased reactive oxygen species (ROS) production. This model suggests that intervention in mitochondrial aging may be possible using appropriate NAD⁺ precursors such as nicotinamide riboside. Restoring NAD⁺ levels may be beneficial throughout the organism. For example, aging-associated disturbances in circadian rhythm are linked to diminished SIRT1 activity, and loss of hematopoietic stem cell function to reduced SIRT3. Work to elucidate other biphasic aging mechanisms is strongly encouraged.
Re: NAD+ for mitochondria
In the following paper discussing the effects of nicotinamide riboside (NR) in a mouse model of Alzheimer's Disease, NR was found to increase NAD+ in the brain.CureOrBust wrote:I did a quick search, but could not find any direct information regarding if this product readily crosses the BBB?
http://www.ncbi.nlm.nih.gov/pubmed/23312803
We found that this NR treatment significantly increased the steady-state levels of NAD+ in the cerebral cortex (Fig. 1B).
Re: NAD+ for mitochondria
The last paragraph in the introduction of the following paper discusses the mechanism of nicotinamide riboside (NR).
http://www.ncbi.nlm.nih.gov/pubmed/24711540
http://www.ncbi.nlm.nih.gov/pubmed/24711540
As such, NAD+ precursors, such as NR, are not equivalent to NADH supplements, such as Panmol NADH, as NR increases the NAD+/NADH ratio activating SIRT1. In contrast, NADH would lower the NAD+/NADH ratio. For now, I recommend against taking NADH.Nutrient sensors Sirt1 and AMPK monitor tissue NAD+/NADH and AMP/ATP ratios, respectively. If these ratios increased, the sensors induce a metabolic fasting response, boosting mitochondrial biogenesis, fatty acid oxidation, and oxidative ATP production (Imai et al, 2000; Rodgers et al, 2005; Canto et al, 2009, 2012). We hypothesized that RCD reduces NADH utilization, decreasing NAD+/NADH and signaling for high nutrient availability, leaving Sirt1 inactive and attenuating mitochondrial biogenesis. However, RCD also decreases ATP production, increasing AMP/ATP, signaling for low nutrition availability, and leading to a potential conflict in nutrient sensor activation and a partial pseudo-starvation response (Nunnari & Suomalainen, 2012). Previous treatment trials of RC-deficient mice have indicated that activation of mitochondrial biogenesis may be beneficial: High-fat diet (Ahola-Erkkila et al, 2010), PPAR (Wenz et al, 2008; Yatsuga & Suomalainen, 2011) and AMPK agonists (Viscomi et al, 2011) all delayed disease progression. Recently, nicotinamide riboside (NR), a vitamin B3-analogue and NAD+ precursor, was shown to promote oxidative metabolism in mice by increasing NAD+/NADH (Bieganowski & Brenner, 2004; Canto et al, 2012). Furthermore, NAD+ precursors improved mitochondrial function in aging mice to mimic that of young mice, in SIRT1-dependent manner (Gomes et al, 2013).
Re: NAD+ for mitochondria
Results from First Human Clinical Study Demonstrate ChromaDex's NIAGEN(R) Nicotinamide Riboside Effectively Increases the Co-enzyme NAD+ and is Safe
https://globenewswire.com/news-release/ ... -Safe.html
https://globenewswire.com/news-release/ ... -Safe.html
IRVINE, Calif., Aug. 12, 2015 (GLOBE NEWSWIRE) -- ChromaDex Corp. (OTCQX:CDXC), an innovator of proprietary health, wellness, and nutritional ingredients that creates science-based solutions for dietary supplements, food and beverage, skin care, sports nutrition, and pharmaceutical products, announced today that results of the first controlled human clinical study on the use of the Company's NIAGEN® nicotinamide riboside (NR) were presented at 4th Federation of American Societies for Experimental Biology (FASEB) Science Research Conference on NAD+ Metabolism and Signaling, currently being held in Germany.
Charles Brenner, PhD, a key member of the ChromaDex Scientific Advisory Board presented an oral platform talk on "How Nicotinamide Riboside Promotes Weight Loss." In addition, Dr. Brenner, Mr. Frank Jaksch, Dr. Ryan Dellinger, and their co-workers presented a poster entitled, "Dose-Dependent Elevation of the Blood NAD Metabolome by NR in Healthy Human Beings."
The oral presentation and poster presented data which indicate that single doses of NIAGEN® NR can elevate the co-enzyme NAD+ in the blood by as much as 2.7-fold. In the first-in-humans clinical trial which involved dosing twelve healthy adult subjects, the group showed that blood cell NAD+ increased with single 100 mg, 300 mg and 1 gram doses of NIAGEN® NR. Average maximal increases in blood NAD+ were approximately 30% at the 100 mg dose and approximately 50% at the higher doses. Increases in blood NAD+ tended to be sustained for longer times at higher doses.
Re: NAD+ for mitochondria
Nicotinamide riboside, an NAD+ precursor, delays axonal degeneration following axotomy [axonal transection].
Stimulation of nicotinamide adenine dinucleotide biosynthetic pathways delays axonal degeneration after axotomy.
J Neurosci. 2006 Aug 16;26(33):8484-91.
Axonal degeneration occurs in many neurodegenerative diseases and after traumatic injury and is a self-destructive program independent from programmed cell death. Previous studies demonstrated that overexpression of nicotinamide mononucleotide adenylyltransferase 1 (Nmnat1) or exogenous application of nicotinamide adenine dinucleotide (NAD) can protect axons of cultured dorsal root ganglion (DRG) neurons from degeneration caused by mechanical or neurotoxic injury. In mammalian cells, NAD can be synthesized from multiple precursors, including tryptophan, nicotinic acid, nicotinamide, and nicotinamide riboside (NmR), via multiple enzymatic steps. To determine whether other components of these NAD biosynthetic pathways are capable of delaying axonal degeneration, we overexpressed each of the enzymes involved in each pathway and/or exogenously administered their respective substrates in DRG cultures and assessed their capacity to protect axons after axotomy. Among the enzymes tested, Nmnat1 had the strongest protective effects, whereas nicotinamide phosphoribosyl transferase and nicotinic acid phosphoribosyl transferase showed moderate protective activity in the presence of their substrates. Strong axonal protection was also provided by Nmnat3, which is predominantly located in mitochondria, and an Nmnat1 mutant localized to the cytoplasm, indicating that the subcellular location of NAD production is not crucial for protective activity. In addition, we showed that exogenous application of the NAD precursors that are the substrates of these enzymes, including nicotinic acid mononucleotide, nicotinamide mononucleotide, and NmR, can also delay axonal degeneration. These results indicate that stimulation of NAD biosynthetic pathways via a variety of interventions may be useful in preventing or delaying axonal degeneration.
Free full text.
Stimulation of nicotinamide adenine dinucleotide biosynthetic pathways delays axonal degeneration after axotomy.
J Neurosci. 2006 Aug 16;26(33):8484-91.
Axonal degeneration occurs in many neurodegenerative diseases and after traumatic injury and is a self-destructive program independent from programmed cell death. Previous studies demonstrated that overexpression of nicotinamide mononucleotide adenylyltransferase 1 (Nmnat1) or exogenous application of nicotinamide adenine dinucleotide (NAD) can protect axons of cultured dorsal root ganglion (DRG) neurons from degeneration caused by mechanical or neurotoxic injury. In mammalian cells, NAD can be synthesized from multiple precursors, including tryptophan, nicotinic acid, nicotinamide, and nicotinamide riboside (NmR), via multiple enzymatic steps. To determine whether other components of these NAD biosynthetic pathways are capable of delaying axonal degeneration, we overexpressed each of the enzymes involved in each pathway and/or exogenously administered their respective substrates in DRG cultures and assessed their capacity to protect axons after axotomy. Among the enzymes tested, Nmnat1 had the strongest protective effects, whereas nicotinamide phosphoribosyl transferase and nicotinic acid phosphoribosyl transferase showed moderate protective activity in the presence of their substrates. Strong axonal protection was also provided by Nmnat3, which is predominantly located in mitochondria, and an Nmnat1 mutant localized to the cytoplasm, indicating that the subcellular location of NAD production is not crucial for protective activity. In addition, we showed that exogenous application of the NAD precursors that are the substrates of these enzymes, including nicotinic acid mononucleotide, nicotinamide mononucleotide, and NmR, can also delay axonal degeneration. These results indicate that stimulation of NAD biosynthetic pathways via a variety of interventions may be useful in preventing or delaying axonal degeneration.
Free full text.
Re: NAD+ for mitochondria
Nicotinamide riboside activates the NAD+ dependent Sirtuin gene SIRT1. SIRT1 activation was found to alleviate experimental optic neuritis in the EAE mouse.
SIRT1 activation confers neuroprotection in experimental optic neuritis.
Invest Ophthalmol Vis Sci. 2007 Aug;48(8):3602-9.
PURPOSE: Axonal damage and loss of neurons correlate with permanent vision loss and neurologic disability in patients with optic neuritis and multiple sclerosis (MS). Current therapies involve immunomodulation, with limited effects on neuronal damage. The authors examined potential neuroprotective effects in optic neuritis by SRT647 and SRT501, two structurally and mechanistically distinct activators of SIRT1, an enzyme involved in cellular stress resistance and survival.
METHODS: Experimental autoimmune encephalomyelitis (EAE), an animal model of MS, was induced by immunization with proteolipid protein peptide in SJL/J mice. Optic neuritis developed in two thirds of eyes with significant retinal ganglion cell (RGC) loss detected 14 days after immunization. RGCs were labeled in a retrograde fashion with fluorogold by injection into superior colliculi. Optic neuritis was detected by inflammatory cell infiltration of the optic nerve.
RESULTS: Intravitreal injection of SIRT1 activators 0, 3, 7, and 11 days after immunization significantly attenuated RGC loss in a dose-dependent manner. This neuroprotective effect was blocked by sirtinol, a SIRT1 inhibitor. Treatment with either SIRT1 activator did not prevent EAE or optic nerve inflammation. A single dose of SRT501 on day 11 was sufficient to limit RGC loss and to preserve axon function.
CONCLUSIONS: SIRT1 activators provide an important potential therapy to prevent the neuronal damage that leads to permanent neurologic disability in optic neuritis and MS patients. Intravitreal administration of SIRT1 activators does not suppress inflammation in this model, suggesting that their neuroprotective effects will be additive or synergistic with current immunomodulatory therapies.
Free full text.
SIRT1 activation confers neuroprotection in experimental optic neuritis.
Invest Ophthalmol Vis Sci. 2007 Aug;48(8):3602-9.
PURPOSE: Axonal damage and loss of neurons correlate with permanent vision loss and neurologic disability in patients with optic neuritis and multiple sclerosis (MS). Current therapies involve immunomodulation, with limited effects on neuronal damage. The authors examined potential neuroprotective effects in optic neuritis by SRT647 and SRT501, two structurally and mechanistically distinct activators of SIRT1, an enzyme involved in cellular stress resistance and survival.
METHODS: Experimental autoimmune encephalomyelitis (EAE), an animal model of MS, was induced by immunization with proteolipid protein peptide in SJL/J mice. Optic neuritis developed in two thirds of eyes with significant retinal ganglion cell (RGC) loss detected 14 days after immunization. RGCs were labeled in a retrograde fashion with fluorogold by injection into superior colliculi. Optic neuritis was detected by inflammatory cell infiltration of the optic nerve.
RESULTS: Intravitreal injection of SIRT1 activators 0, 3, 7, and 11 days after immunization significantly attenuated RGC loss in a dose-dependent manner. This neuroprotective effect was blocked by sirtinol, a SIRT1 inhibitor. Treatment with either SIRT1 activator did not prevent EAE or optic nerve inflammation. A single dose of SRT501 on day 11 was sufficient to limit RGC loss and to preserve axon function.
CONCLUSIONS: SIRT1 activators provide an important potential therapy to prevent the neuronal damage that leads to permanent neurologic disability in optic neuritis and MS patients. Intravitreal administration of SIRT1 activators does not suppress inflammation in this model, suggesting that their neuroprotective effects will be additive or synergistic with current immunomodulatory therapies.
Free full text.
Re: NAD+ for mitochondria
Additive or synergistic is especially good.NHE wrote: CONCLUSIONS: SIRT1 activators provide an important potential therapy to prevent the neuronal damage that leads to permanent neurologic disability in optic neuritis and MS patients. Intravitreal administration of SIRT1 activators does not suppress inflammation in this model, suggesting that their neuroprotective effects will be additive or synergistic with current immunomodulatory therapies.
Re: NAD+ for mitochondria
Nicotinamide riboside is uniquely and orally bioavailable in mice and humans
Nature Communications. 2016 Oct 10;7:12948.
Nicotinamide riboside (NR) is in wide use as an NAD+ precursor vitamin. Here we determine the time and dose-dependent effects of NR on blood NAD+ metabolism in humans. We report that human blood NAD+ can rise as much as 2.7-fold with a single oral dose of NR in a pilot study of one individual, and that oral NR elevates mouse hepatic NAD+ with distinct and superior pharmacokinetics to those of nicotinic acid and nicotinamide. We further show that single doses of 100, 300 and 1,000 mg of NR produce dose-dependent increases in the blood NAD+ metabolome in the first clinical trial of NR pharmacokinetics in humans. We also report that nicotinic acid adenine dinucleotide (NAAD), which was not thought to be en route for the conversion of NR to NAD+, is formed from NR and discover that the rise in NAAD is a highly sensitive biomarker of effective NAD+ repletion.
Free full text
Nature Communications. 2016 Oct 10;7:12948.
Nicotinamide riboside (NR) is in wide use as an NAD+ precursor vitamin. Here we determine the time and dose-dependent effects of NR on blood NAD+ metabolism in humans. We report that human blood NAD+ can rise as much as 2.7-fold with a single oral dose of NR in a pilot study of one individual, and that oral NR elevates mouse hepatic NAD+ with distinct and superior pharmacokinetics to those of nicotinic acid and nicotinamide. We further show that single doses of 100, 300 and 1,000 mg of NR produce dose-dependent increases in the blood NAD+ metabolome in the first clinical trial of NR pharmacokinetics in humans. We also report that nicotinic acid adenine dinucleotide (NAAD), which was not thought to be en route for the conversion of NR to NAD+, is formed from NR and discover that the rise in NAAD is a highly sensitive biomarker of effective NAD+ repletion.
Free full text
Re: NAD+ for mitochondria
For what it's worth, I tried using nicotinamide riboside (NR) for about 6½ months. For the first 3 months I used Life Extension's NR. I then switched to Thorne Research's NR since it was about 33% less expensive on a $/mg basis. Near the end of that 5 month period I started getting bad chest cramps that only got worse with each passing day. I hypothesized that it was related to the NR since that was the only thing that I had changed and that it may have been due to the less expensive NR from Thorne so I stopped taking it. I felt better after a couple of days. I started back up on the Life Extension brand a few weeks later. The chest cramps returned after 6 weeks. I stopped taking it altogether. Yes, I've read several of the papers on it and was impressed by the changes in mitochondria, e.g., increased mitochondrial numbers and increased cristae content. However, until I find some explanation for the chest cramps and a viable solution for avoiding them, I'm not going back on NR.
Your experience may, of course, be different from mine.
Your experience may, of course, be different from mine.
-
grandsons4
- Family Member
- Posts: 87
- Joined: Wed Aug 14, 2013 6:43 pm
Re: NAD+ for mitochondria
Would sulforaphane suffice to replace NR?
Re: NAD+ for mitochondria
Only time will tell if the supplement will be of benefit, after controlled trials on humans are completed. And even then, we may not know for sure whether this B3 supplement can deliver some of the hefty promises its makers makes. They (the manufacturer Chromadex) have obviously made a tremendous investment in this and will obviously do its best to market it.
If you take a look at the reviews of the numerous Niagen product reviews on Amazon, (there are probably around a dozen), the results are mixed. Of course, these reviews are far from scientific and there are many fake reviews on Amaazon.
As this supplement has been available for a number of years and there is not as of now definitive evidence of its abilities, I am suspect. But I take a lot of supplements and have still decided to add this one. Because you never know!
If you take a look at the reviews of the numerous Niagen product reviews on Amazon, (there are probably around a dozen), the results are mixed. Of course, these reviews are far from scientific and there are many fake reviews on Amaazon.
As this supplement has been available for a number of years and there is not as of now definitive evidence of its abilities, I am suspect. But I take a lot of supplements and have still decided to add this one. Because you never know!
Re: NAD+ for mitochondria
Sulforaphane is an isothiocyanate.grandsons4 wrote:Would sulforaphane suffice to replace NR?
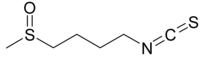
I don't know if it's biological activity can substitute for nicotinamide riboside which has a very different structure.

One activity of sulforaphane is to activate Nrf2.
Here's something interesting. Nrf2 activity supports mitochondrial function.
The multifaceted role of Nrf2 in mitochondrial function.
Curr Opin Toxicol. 2016 Dec;1:80-91.
The transcription factor nuclear factor erythroid 2 p45-related factor 2 (Nrf2) is the master regulator of the cellular redox homeostasis. Nrf2 target genes comprise of a large network of antioxidant enzymes, proteins involved in xenobiotic detoxification, repair and removal of damaged proteins, inhibition of inflammation, as well as other transcription factors. In recent years it has emerged that as part of its role as a regulator of cytoprotective gene expression, Nrf2 impacts mitochondrial function. Increased Nrf2 activity defends against mitochondrial toxins. Reduced glutathione, the principal small molecule antioxidant in the mammalian cell and a product of several of the downstream target genes of Nrf2, counterbalances mitochondrial ROS production. The function of Nrf2 is suppressed in mitochondria-related disorders, such as Parkinson's disease and Friedrich's ataxia. Studies using isolated mitochondria and cultured cells have demonstrated that Nrf2 deficiency leads to impaired mitochondrial fatty acid oxidation, respiration and ATP production. Small molecule activators of Nrf2 support mitochondrial integrity by promoting mitophagy and conferring resistance to oxidative stress-mediated permeability transition. Excitingly, recent studies have shown that Nrf2 also affects mitochondrial function in stem cells with implications for stem cell self-renewal, cardiomyocyte regeneration, and neural stem/progenitor cell survival.
Free full text.
So it looks like both molecules support mitochondrial function, but in different ways.
