The main idea is that there are four patterns of MS (one of them, IV very rare and often ignored), and they represent different pathogenic kinds of MS. Probably the most common, pattern II, is related to EBV while the rest are not. This paper says that there is an autoantibody signature for them.
Antibody signatures in patients with histopathologically defined multiple sclerosis patterns.
https://www.ncbi.nlm.nih.gov/pubmed/31950335
Abstract
Early active multiple sclerosis (MS) lesions can be classified histologically into three main immunopathological patterns of demyelination (patterns I-III), which suggest pathogenic heterogeneity and may predict therapy response. Patterns I and II show signs of immune-mediated demyelination, but only pattern II is associated with antibody/complement deposition. In pattern III lesions, which include Baló's concentric sclerosis, primary oligodendrocyte damage was proposed. Serum antibody reactivities could reflect disease pathogenesis and thus distinguish histopathologically defined MS patterns. We established a customized microarray with more than 700 peptides that represent human and viral antigens potentially relevant for inflammatory demyelinating CNS diseases, and tested sera from 66 patients (pattern I n = 12; II n = 29; III n = 25, including 8 with Baló's), healthy controls, patients with Sjögren's syndrome and stroke patients. Cell-based assays were performed for aquaporin 1 (AQP1) and AQP4 antibody detection. No single peptide showed differential binding among study cohorts. Because antibodies can react with different peptides from one protein, we also analyzed groups of peptides. Patients with pattern II showed significantly higher reactivities to Nogo-A peptides as compared to patterns I (p = 0.02) and III (p = 0.02). Pattern III patients showed higher reactivities to AQP1 (compared to pattern I p = 0.002, pattern II p = 0.001) and varicella zoster virus (VZV, compared to pattern II p = 0.05). In patients with Baló's, AQP1 reactivity was also significantly higher compared to patients without Baló's (p = 0.04), and the former revealed distinct antibody signatures. Histologically, Baló's patients showed loss of AQP1 and AQP4 in demyelinating lesions, but no antibodies binding conformational AQP1 or AQP4 were detected. In summary, higher reactivities to Nogo-A peptides in pattern II patients could be relevant for enhanced axonal repair and remyelination. Higher reactivities to AQP1 peptides in pattern III patients and its subgroup of Baló's patients possibly reflect astrocytic damage. Finally, latent VZV infection may cause peripheral immune activation.
There is also a review here:
https://multiple-sclerosis-research.org ... t-targets/
Additional information (from other papers):
A description of the four patterns is available here:
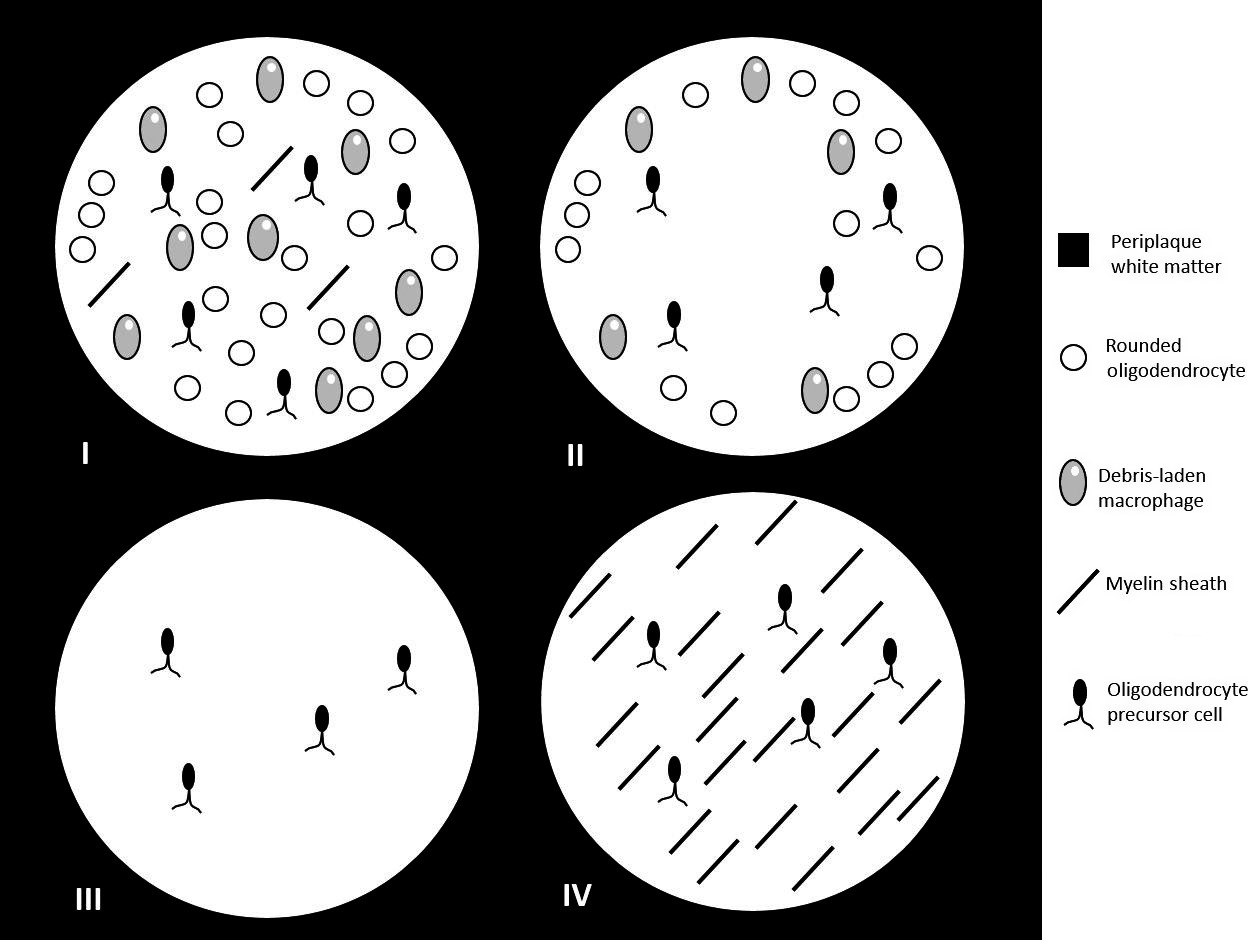
(Image taken from:Imaging in multiple sclerosis: A new spin on lesions, https://onlinelibrary.wiley.com/doi/ful ... 9485.12498)
As of 2018 it seems that pattern IV is currently considered outside the MS spectrum. The three remaining patterns evolve in a similar way and all the three can yield smouldering lesions. It is currently unknown what determines the evolution of a lesion. A better picture of the three remaining patterns could be this one:

(Image taken from Reich et al NEJM 2018, linked through https://multiple-sclerosis-research.org ... ign-in-ms/ )